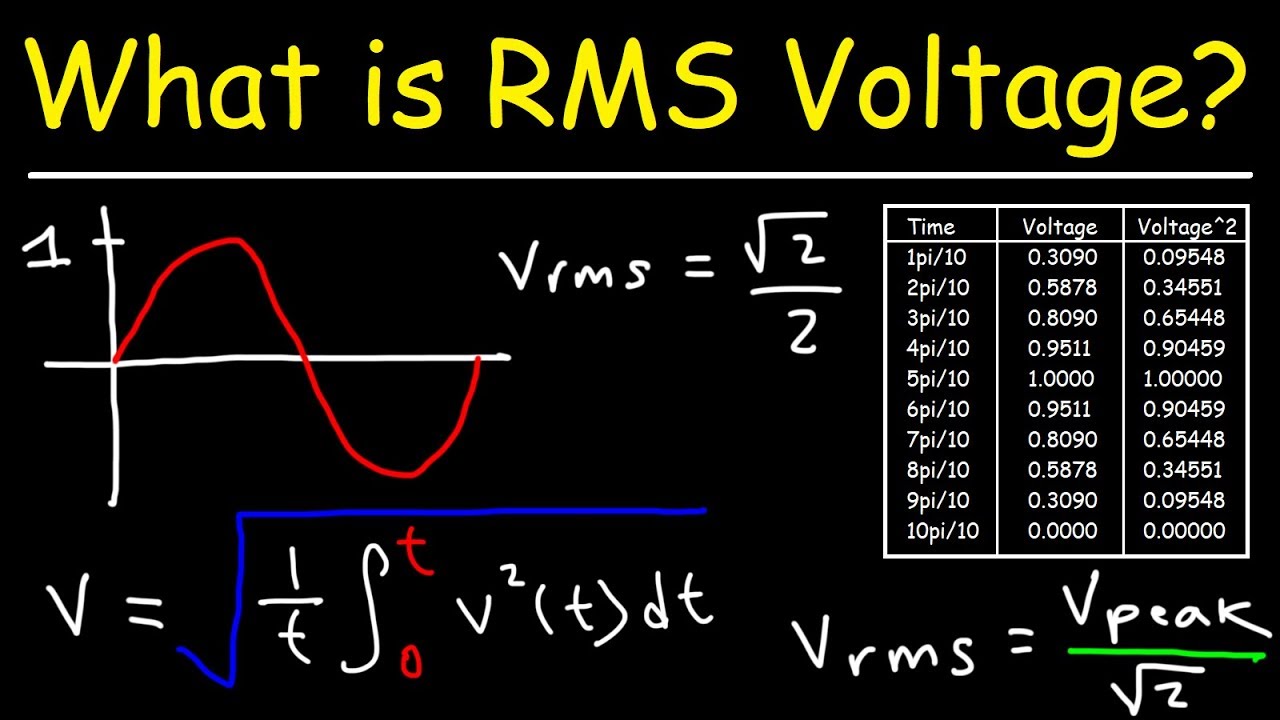
Calculate the rms velocity of molecules of a gas of density 1.5 g litre^(-1) at a pressure of 2 xx 10^(6) N//m^(2).
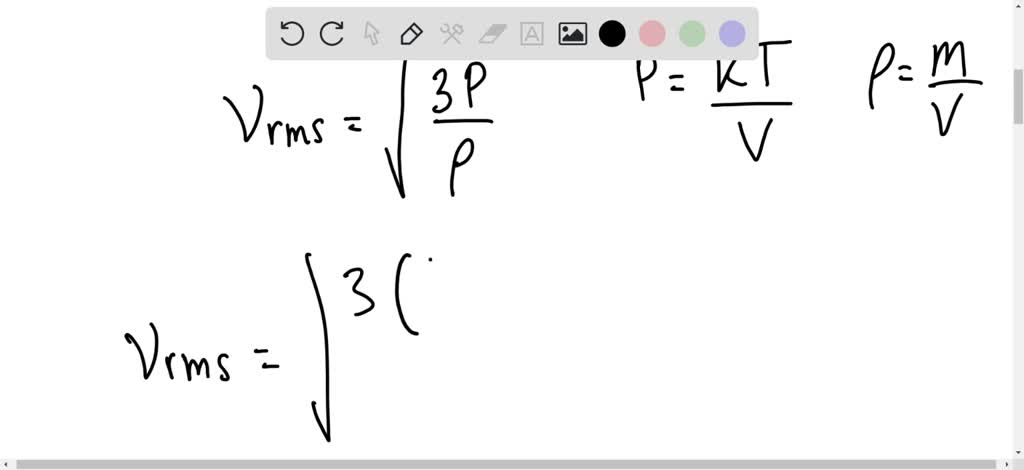
SOLVED: Calculate the root mean square velocity of ammonia (NH3) molecules at 25°C. The molar mass of NH3 is 17.02 g/mol and 1 J = 1 kg m2/s2

RMS Value, Average Value, Peak Value, Peak Factor, Form Factor in AC | Rms, Engineering notes, Electrical circuit diagram



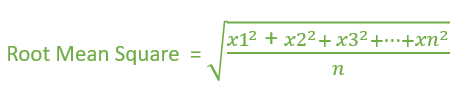
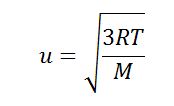
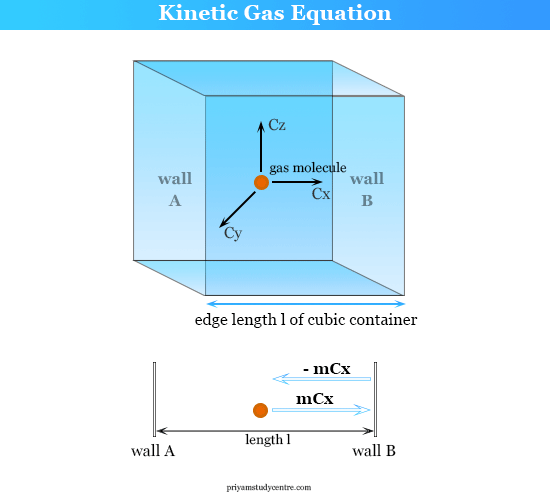




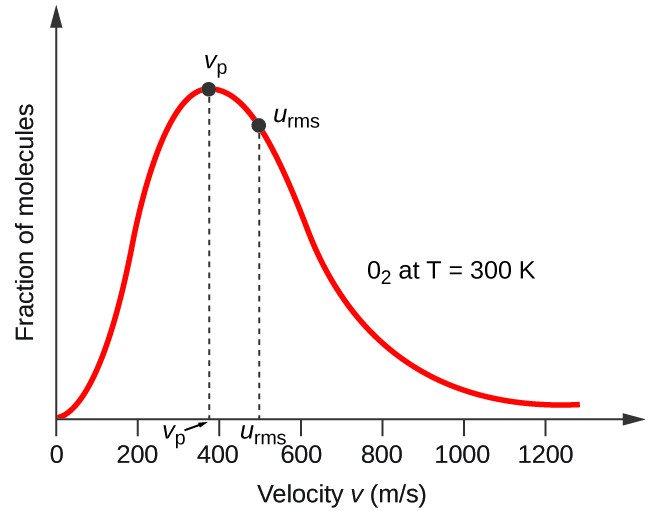

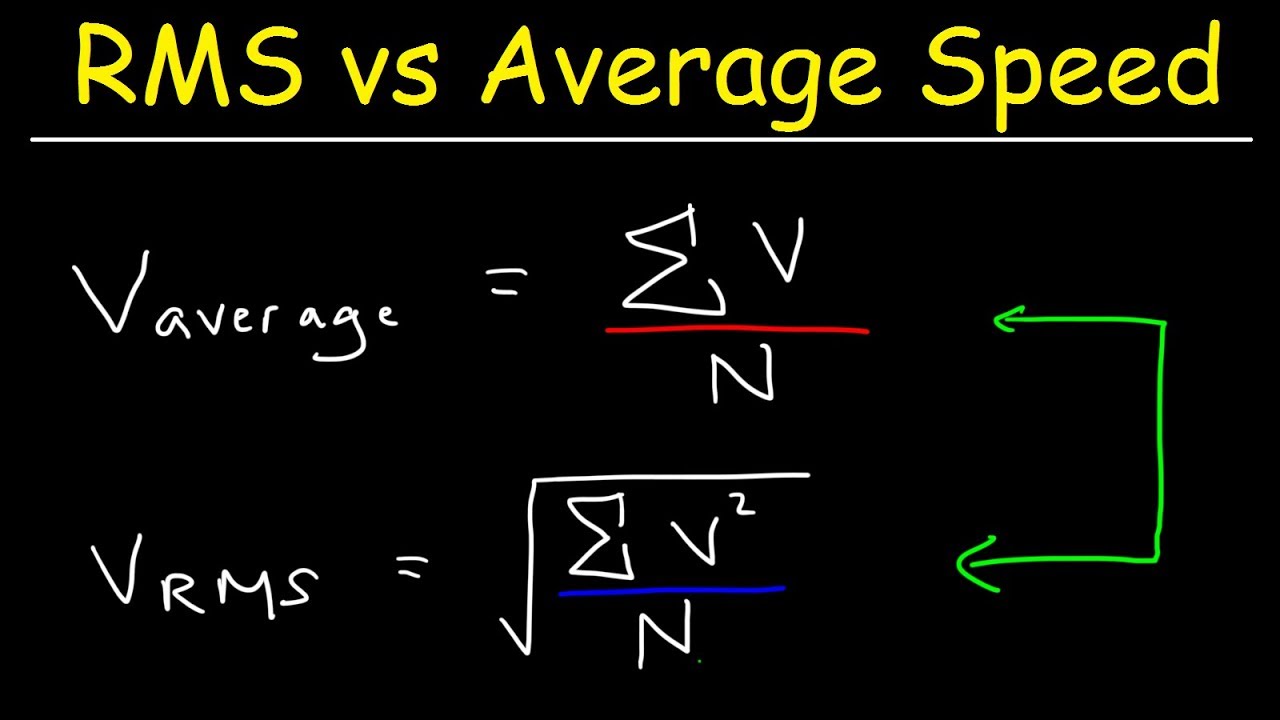


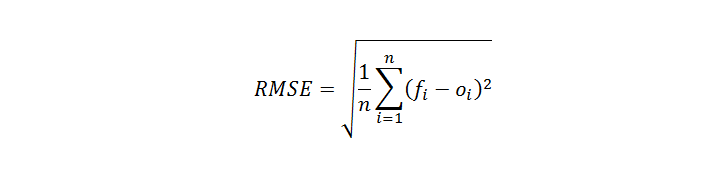


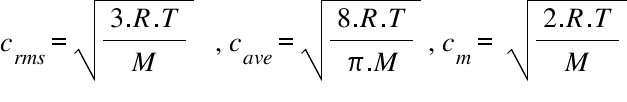


:max_bytes(150000):strip_icc()/ChalkboardCalculations-58b1c7c43df78cdcd8148e15.jpg)