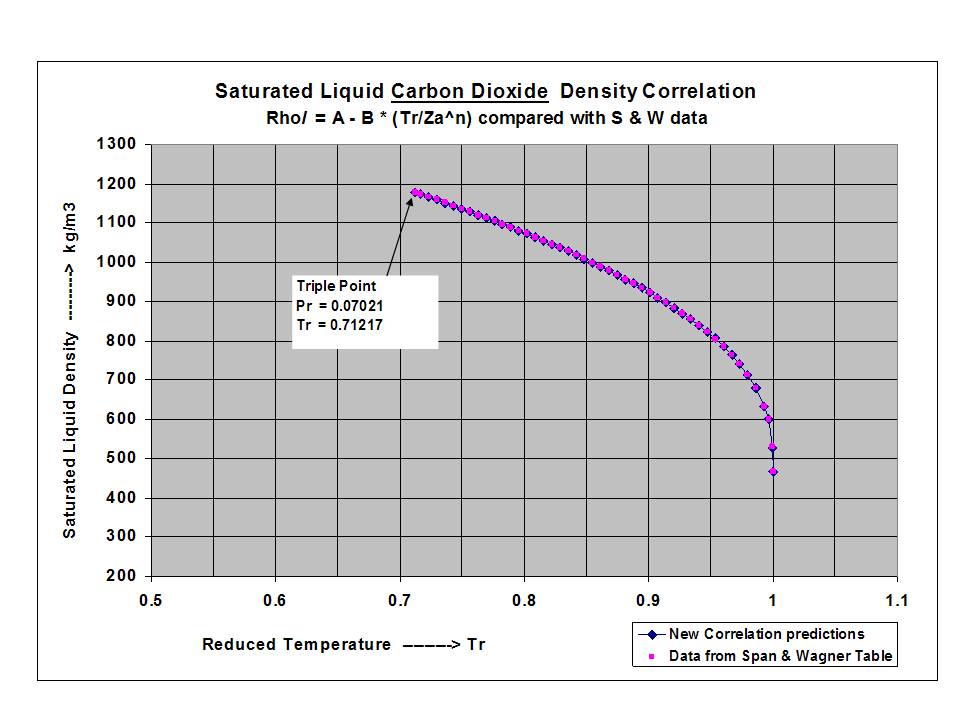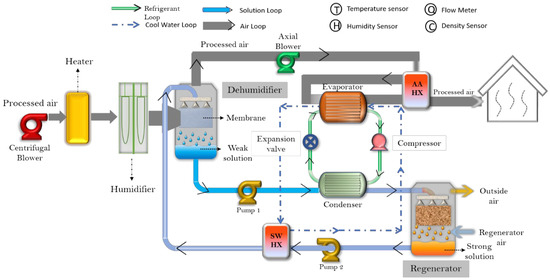
Sustainability | Free Full-Text | Economic and Experimental Assessment of KCOOH Hybrid Liquid Desiccant-Vapor Compression System
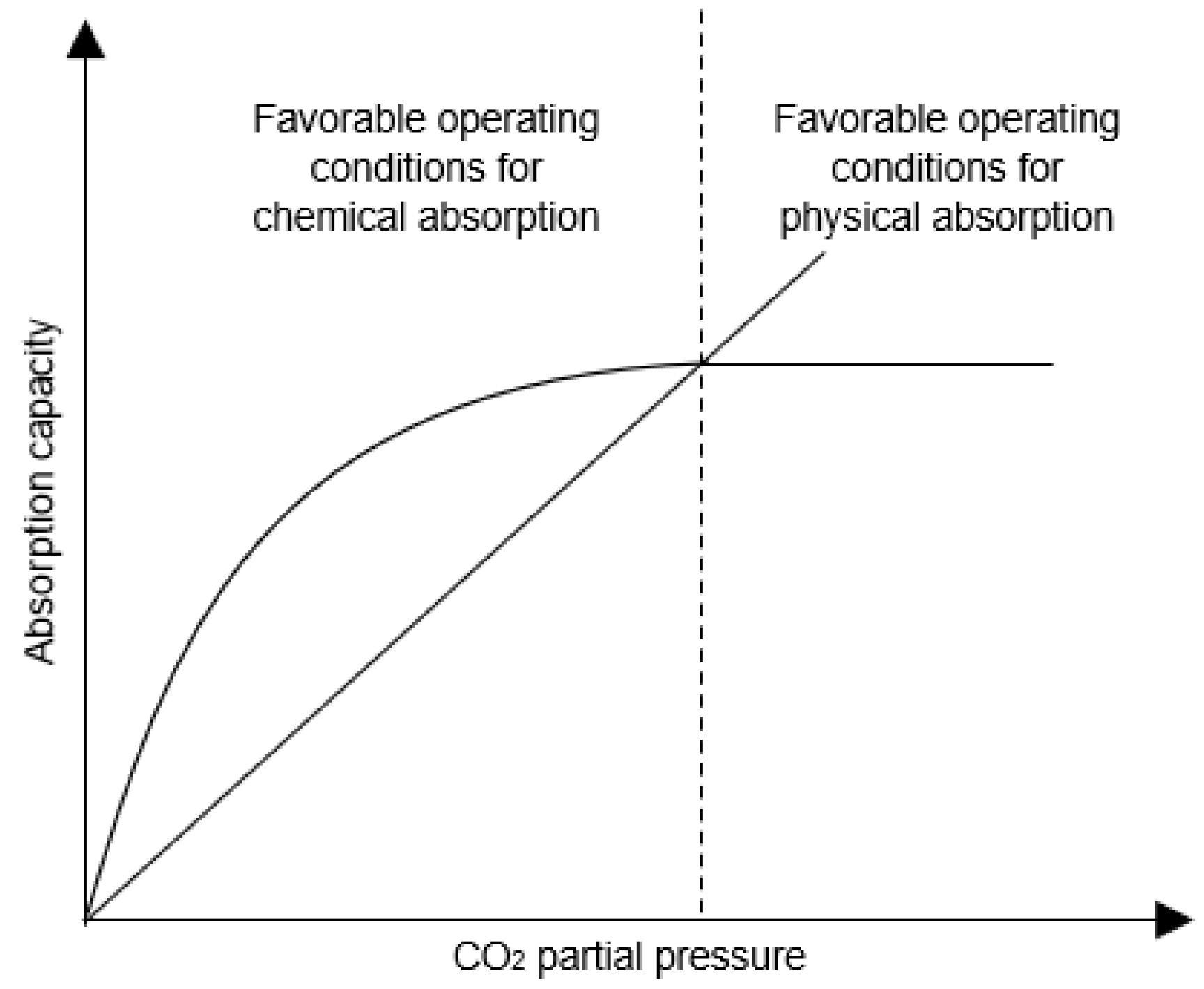
Sustainability | Free Full-Text | Emerging Solvent Regeneration Technologies for CO2 Capture through Offshore Natural Gas Purification Processes

A review of recent trends and emerging perspectives of ionic liquid membranes for CO2 separation - ScienceDirect
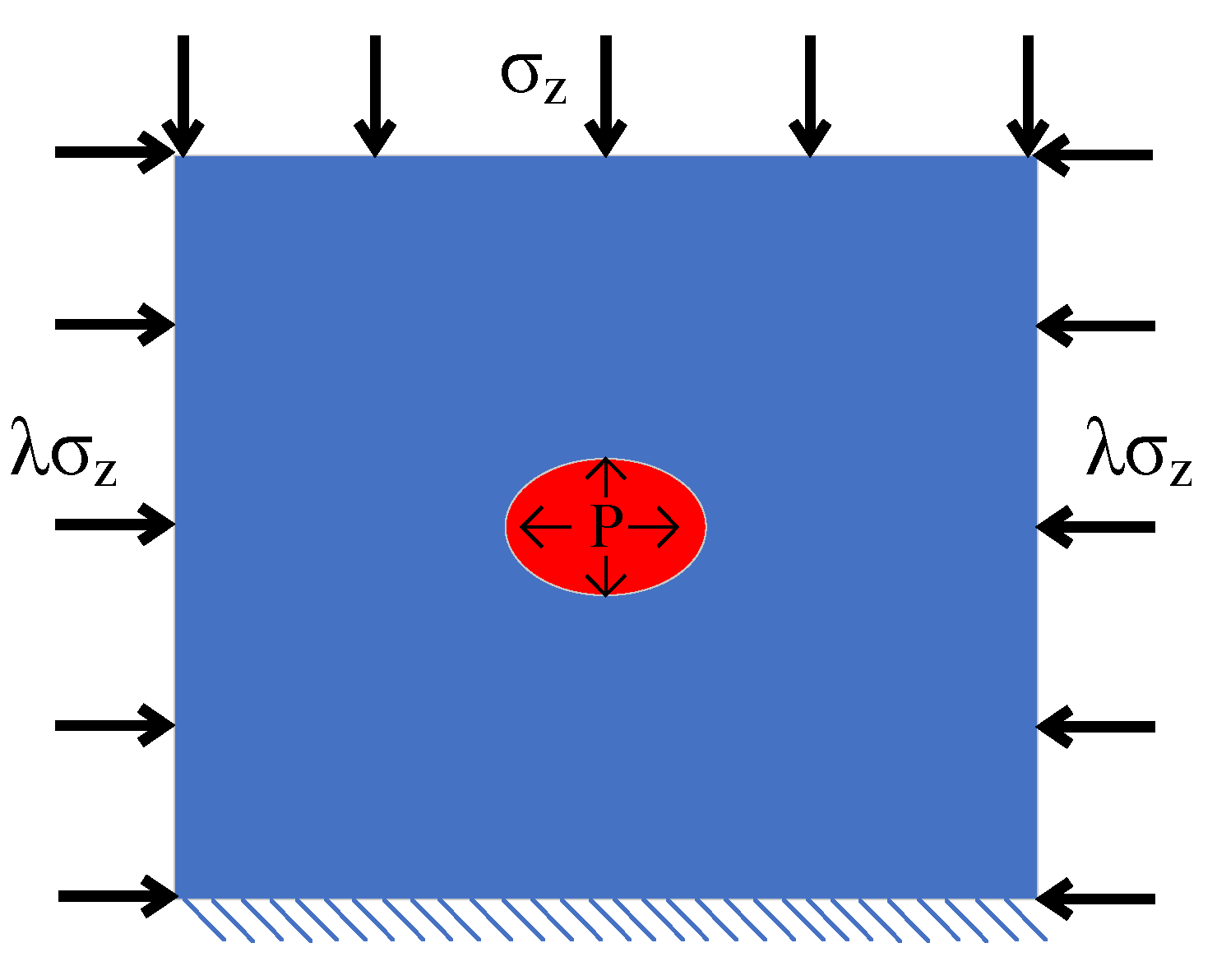
Sustainability | Free Full-Text | Permeability-Enhancing Technology through Liquid CO2 Fracturing and Its Application
![PDF] Vapor-liquid equilibrium data for the carbon dioxide and nitrogen (CO2+N2) system at the temperatures 223, 270, 298 and 303 K and pressures up to 18 MPa | Semantic Scholar PDF] Vapor-liquid equilibrium data for the carbon dioxide and nitrogen (CO2+N2) system at the temperatures 223, 270, 298 and 303 K and pressures up to 18 MPa | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a22254d7b51e24f9e2e2bbbdec04a8c73b9f8e75/10-Figure4-1.png)
PDF] Vapor-liquid equilibrium data for the carbon dioxide and nitrogen (CO2+N2) system at the temperatures 223, 270, 298 and 303 K and pressures up to 18 MPa | Semantic Scholar
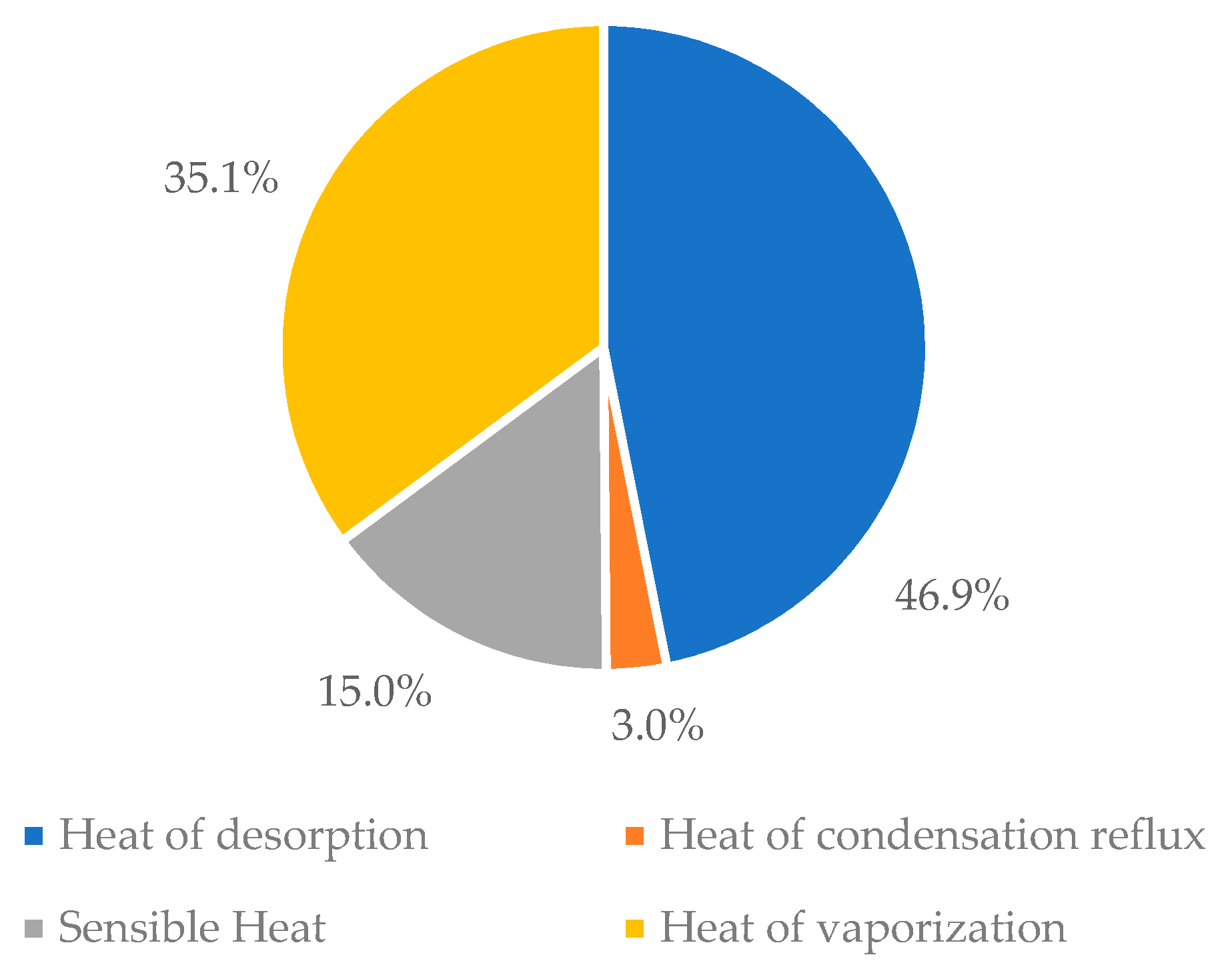
Sustainability | Free Full-Text | Energy Minimization in Piperazine Promoted MDEA-Based CO2 Capture Process
![PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/41236103d2b9c81bab8dfe341d7be95b4e6fb18d/22-Figure2-1.png)
PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar
![PDF] Vapor-liquid equilibrium data for the carbon dioxide and nitrogen (CO2+N2) system at the temperatures 223, 270, 298 and 303 K and pressures up to 18 MPa | Semantic Scholar PDF] Vapor-liquid equilibrium data for the carbon dioxide and nitrogen (CO2+N2) system at the temperatures 223, 270, 298 and 303 K and pressures up to 18 MPa | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a22254d7b51e24f9e2e2bbbdec04a8c73b9f8e75/17-Table10-1.png)
PDF] Vapor-liquid equilibrium data for the carbon dioxide and nitrogen (CO2+N2) system at the temperatures 223, 270, 298 and 303 K and pressures up to 18 MPa | Semantic Scholar

Porous Supramolecular Assembly of Pentiptycene-Containing Gold(I) Complexes: Persistent Excited-State Aurophilicity and Inclusion-Induced Emission Enhancement | Inorganic Chemistry
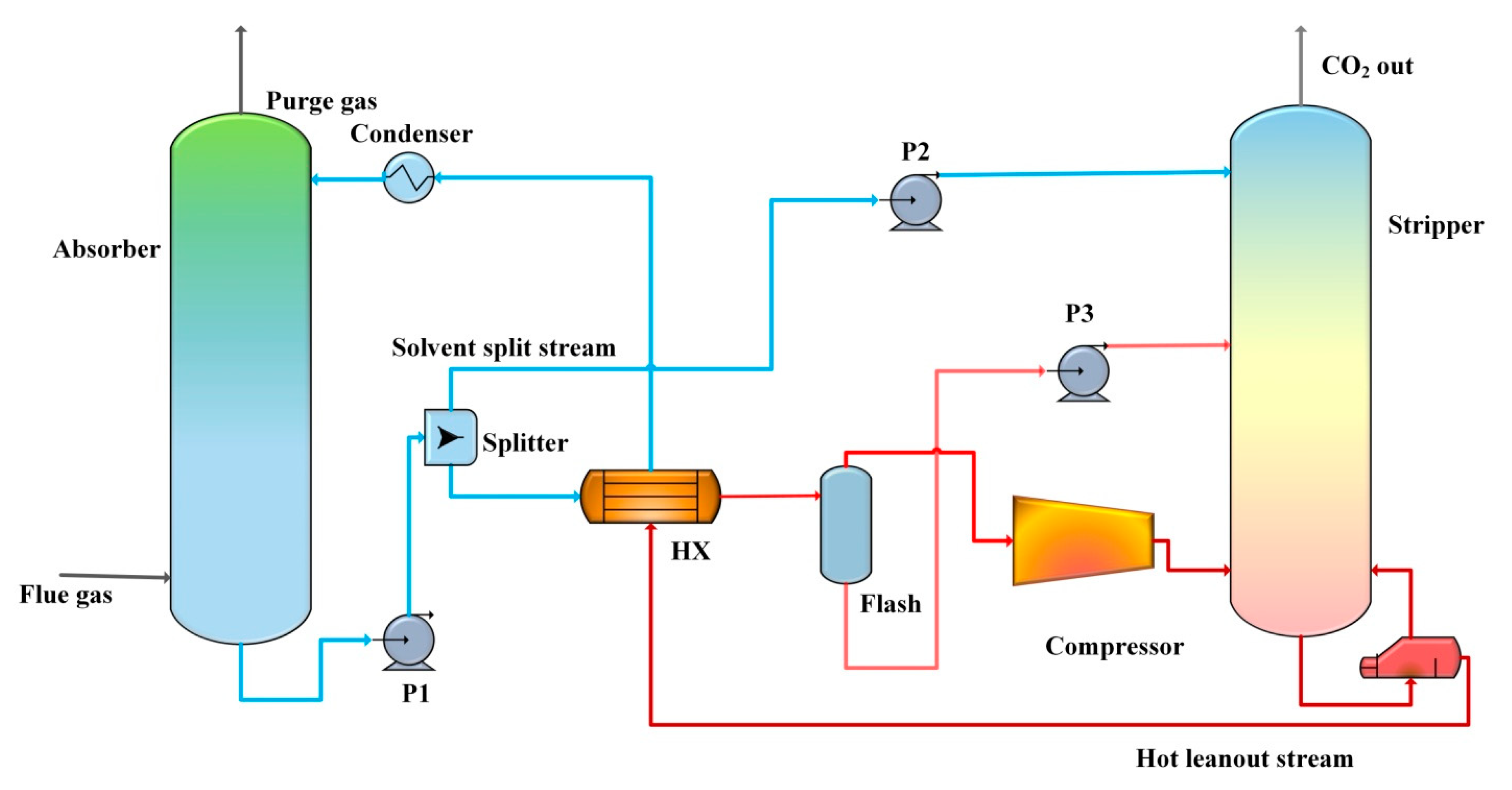
Sustainability | Free Full-Text | A Review on Enhancing Solvent Regeneration in CO2 Absorption Process Using Nanoparticles

Sustainability | Free Full-Text | Evaluation of Decarbonization Technologies for ASEAN Countries via an Integrated Assessment Tool

Important Aspects Regarding the Chemical Stability of Aqueous Amine Solvents for CO2 Capture | Industrial & Engineering Chemistry Research
![PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/41236103d2b9c81bab8dfe341d7be95b4e6fb18d/13-Table2-1.png)
PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar
![PDF] Vapor-liquid equilibrium data for the carbon dioxide and nitrogen (CO2+N2) system at the temperatures 223, 270, 298 and 303 K and pressures up to 18 MPa | Semantic Scholar PDF] Vapor-liquid equilibrium data for the carbon dioxide and nitrogen (CO2+N2) system at the temperatures 223, 270, 298 and 303 K and pressures up to 18 MPa | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a22254d7b51e24f9e2e2bbbdec04a8c73b9f8e75/23-Figure11-1.png)
PDF] Vapor-liquid equilibrium data for the carbon dioxide and nitrogen (CO2+N2) system at the temperatures 223, 270, 298 and 303 K and pressures up to 18 MPa | Semantic Scholar

Simultaneous measurement of the density and viscosity for n-Decane + CO2 binary mixtures at temperature between (303.15 to 373.15) K and pressures up to 80 MPa - ScienceDirect

PDF) Vapor-liquid equilibrium measurements of the binary mixtures CO2 + acetone and CO2 + pentanones
![PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/41236103d2b9c81bab8dfe341d7be95b4e6fb18d/21-Figure1-1.png)
PDF] Vapor–Liquid Equilibrium Data for the Carbon Dioxide (CO2) + 1,1,1,3,3-Pentafluorobutane (R365mfc) System at Temperatures from 283.15 to 337.15 K | Semantic Scholar

PDF) Vapor-liquid equilibrium measurements of the binary mixtures CO2 + acetone and CO2 + pentanones
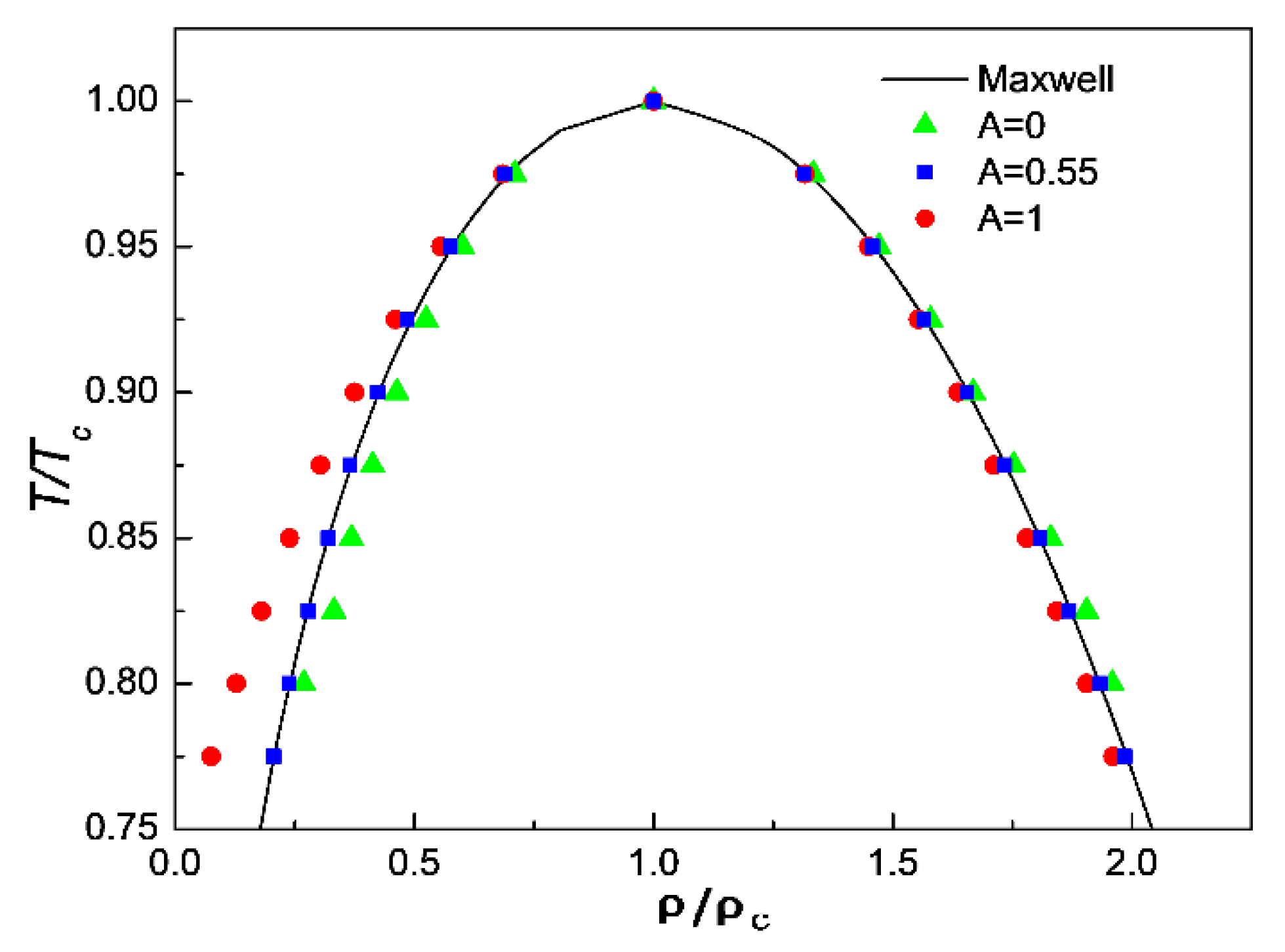
Sustainability | Free Full-Text | Study on Interparticle Interaction Force Model to Correct Saturation Density of Real Cryogenic Fluid for LBM Simulation

Properties and Promise of Catenated Nitrogen Systems As High-Energy-Density Materials | Chemical Reviews